Plasma seems to have the kinds of properties one would like for life. It’s somewhat like liquid water — unpredictable and thus able to behave in an enormously complex fashion. It could probably carry as much information as DNA does. It has at least the potential for organizing itself in interesting ways.
– Freeman Dyson (1986)

Plasma – colloquially referred to as the `4th state of matter’ – exhibits many interesting phenomena due to the diverse range of physics that govern its behavior, including electromagnetics, fluid/statistical mechanics, and even quantum mechanics. Most are familiar with the states of matter that are common on Earth. The reader is guaranteed to have observed how one type of matter in particular – water – transitions between the three common states of matter (solid ice, liquid water, water vapor), and thus they are familiar with the characteristics of each of those phases. A gas is best understood as a collection of molecules that have a high enough average speed that they overcome the intermolecular forces that make them stick to other alike molecules and fly about, experiencing collisions with one another. Plasma occurs when we inject even more energy into these molecules, breaking up their atomic structure in the process.

To proceed with our water example, if we continue to heat up the gas, eventually the covalent bonds that hold together the water molecules will break apart into monatomic particles (the H-O bond energy of water is ![]() 450 kJ/mol, and O
450 kJ/mol, and O![]() has a bond energy of
has a bond energy of ![]() 500 kJ/mol). At this point, the substance is still a gas, albeit a very hot one. As we continue to inject energy into the system, we excite various elevated electronic energy states. and eventually, the electrons in the oxygen and hydrogen molecules will be knocked free of their orbitals (the ionization energies of oxygen and hydrogen are
500 kJ/mol). At this point, the substance is still a gas, albeit a very hot one. As we continue to inject energy into the system, we excite various elevated electronic energy states. and eventually, the electrons in the oxygen and hydrogen molecules will be knocked free of their orbitals (the ionization energies of oxygen and hydrogen are ![]() 1200 kJ/mol), and we are left with a plasma. Although this state of matter is uncommon on Earth (mostly observed by the naked eye in the form of lightning/electrical arcs and the aurora borealis), it is actually the state of matter that is most common in the universe as it is the phase of matter that stars are composed of. In the figure below, we see an oft-reproduced figure in the world of plasma physics, a plot of temperature vs. density, with photos of examples of plasmas that exist within the regime of density and temperature at that location in the plot. As one can see from this figure, plasma exists in the cold expanse of interstellar space, within comet tails, stellar nebulae, stars, lightning, and in fusion devices like those pictured at the top of the figure (Sandia Z-machine on the left, and an inertial confinement hohlraum on the right). Below what is called the Fermi temperature (a statistical/quantum mechanics-derived concept that arises from the statistical properties of some types of fundamental particles, here denoted by a solid black line), solid-state plasmas exist, which can be found in the cores of stars, and even in the form of metals here on Earth, which themselves are best-modeled as a plasma due to the fact that the electrons within them are more or less free to move.
1200 kJ/mol), and we are left with a plasma. Although this state of matter is uncommon on Earth (mostly observed by the naked eye in the form of lightning/electrical arcs and the aurora borealis), it is actually the state of matter that is most common in the universe as it is the phase of matter that stars are composed of. In the figure below, we see an oft-reproduced figure in the world of plasma physics, a plot of temperature vs. density, with photos of examples of plasmas that exist within the regime of density and temperature at that location in the plot. As one can see from this figure, plasma exists in the cold expanse of interstellar space, within comet tails, stellar nebulae, stars, lightning, and in fusion devices like those pictured at the top of the figure (Sandia Z-machine on the left, and an inertial confinement hohlraum on the right). Below what is called the Fermi temperature (a statistical/quantum mechanics-derived concept that arises from the statistical properties of some types of fundamental particles, here denoted by a solid black line), solid-state plasmas exist, which can be found in the cores of stars, and even in the form of metals here on Earth, which themselves are best-modeled as a plasma due to the fact that the electrons within them are more or less free to move.

Now we can start to get more precise. First, let’s get an idea of how much ionization is present in a given gas at a given temperature. Suppose we want to figure out an approximation for what fraction of air molecules are ionized. Let’s simplify our analysis by assuming that air is roughly equivalent to monatomic nitrogen, and then let’s find our how much of it is singly-ionized. If we assume we are in thermal equilibrium at a temperature that is not close to zero, then we are safe in assuming that the energy distribution of the particles follows the Boltzmann distribution (see the derivation in another learn page) where the number of particles in the ![]() th energy level is
th energy level is ![]() , where
, where ![]() is the number of available states in the
is the number of available states in the ![]() th energy level,
th energy level, ![]() is the
is the ![]() th energy level relative to the ground state and
th energy level relative to the ground state and ![]() is the partition function [1]. If we sum over all the energy levels to find the total number of particles in each species (the three species here are neutral atomic nitrogen, singly ionized nitrogen N
is the partition function [1]. If we sum over all the energy levels to find the total number of particles in each species (the three species here are neutral atomic nitrogen, singly ionized nitrogen N![]() , and electrons) and compute the quotient of these totals, we get an expression reminiscent of the Law of Mass Action:
, and electrons) and compute the quotient of these totals, we get an expression reminiscent of the Law of Mass Action:
![]()
where ![]() is the ionization energy.
is the ionization energy.
The partition function in general has two contributions, translational and internal; ![]() . For the electrons, the internal contribution is simply
. For the electrons, the internal contribution is simply ![]() since they are fundamental particles that can either be spin up or spin down. The internal contributions of the atoms and ions are more complicated and have to do with electronic excitation (since we are dealing with monatomic gas, we do not need to concern ourselves with rotation and vibration of molecular bonds), but are mostly second order so we will assume
since they are fundamental particles that can either be spin up or spin down. The internal contributions of the atoms and ions are more complicated and have to do with electronic excitation (since we are dealing with monatomic gas, we do not need to concern ourselves with rotation and vibration of molecular bonds), but are mostly second order so we will assume ![]() for both. The translational contribution to the partition function is, in general,
for both. The translational contribution to the partition function is, in general,
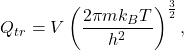
where ![]() J-s is Planck’s constant. This partition function arises from considering the available translational energy states and assuming we are in the Boltzmann limit (states sparsely populated). Now, we need to note a couple of things. First,
J-s is Planck’s constant. This partition function arises from considering the available translational energy states and assuming we are in the Boltzmann limit (states sparsely populated). Now, we need to note a couple of things. First, ![]() . Second, we can replace the volume term
. Second, we can replace the volume term ![]() with
with ![]() , where
, where ![]() is the number density of the given species. Finally, since each ionization event creates exactly one ion and one electron, we have
is the number density of the given species. Finally, since each ionization event creates exactly one ion and one electron, we have ![]() . With these things in mind, we get:
. With these things in mind, we get:
(1) 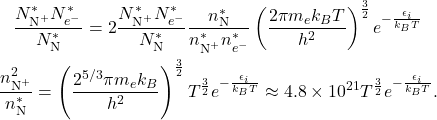
This equation is referred to as the Saha equation, and gives us an estimate for degree of ionization of a gas in thermal equilibrium. For our nitrogen example, if we assume we are at room temperature ![]() 300 K where the neutral density is about
300 K where the neutral density is about ![]() m
m![]() , since the ionization energy of nitrogen is
, since the ionization energy of nitrogen is ![]() eV, the fractional ionization is basically nil;
eV, the fractional ionization is basically nil; ![]() . That matches our experience; we do not see much plasma appearing at sea level. But nonetheless, the Saha equation tells us that every gas has some degree of ionization, so where do we draw the line and call it a plasma?
. That matches our experience; we do not see much plasma appearing at sea level. But nonetheless, the Saha equation tells us that every gas has some degree of ionization, so where do we draw the line and call it a plasma?
Quantitative and Qualitative Descriptions and Criteria
The definition of plasma given in [2] is that plasma is a quasineutral gas of charges and neutral particles which exhibits collective behavior. Collective behavior here is a bit of a qualitative term; it is referring to the characteristic of plasma that Dyson was describing in the epigraph preceding this section. By collective behavior, we mean that local behavior of the constituents of a plasma is driven by the motion of other constituents, sometimes quite strongly. Plasma gets its name from Irving Langmuir who was reminded of the properties of blood plasma, but it’s a bit of a misnomer since plasma translates in Ancient Greek to moldable `substance’, where plasma is anything but. If we `squeeze’ a plasma in one location, we create some sort of strong local current density and displacement, which then produces the resultant EM fields, which then go on to affect the motion of the constituents of the plasma both near and far from our `squeeze’ location. Because of this, plasma seems to have a mind of its own. Moreover, if the degree of ionization is too low, there is not enough charge motion and separation to create the requisite macroscopic fields to strongly affect particles far away from the `squeeze’.
The `quasi-neutral’ part of the definition has to do with a phenomenon called `shielding’. Imagine we have a plasma background with a decently high density (forgive the imprecise language; I will put a quantitative criterion on this shortly), and then we place a positive test charge in the plasma. Immediately, the electrons in the plasma will rush to meet the test charge, that is, until the ions that they `left behind’ (ions are much more massive than electrons and thus their inertia prevents them from moving on the same time scale) begin to represent an effective cloud of positive charge surrounding the system, pulling the electrons back in the direction from which they came. If one draws a boundary around this area of charge displacement, the view from outside the boundary is as if no test charge is present since the constituents of the plasma have shielded out its potential. If there was no plasma present, an observer form the same distance would see the potential produced by the test charge in vacuum. A diagram of this scenario is shown in the figure below.

We’re going to derive the length scale over which this shielding occurs. Let’s imagine we have a plane of constant potential ![]() at
at ![]() in an infinite uniform singly ionized plasma, and then let’s see how the potential changes as we move away from the origin. To keep things simple, we ignore the ion motion due to the mismatch in inertia I mentioned before, so there is a uniform background of positive ions in which electrons are free to move about. In this case, Poisson’s equation (Gauss’ law with
in an infinite uniform singly ionized plasma, and then let’s see how the potential changes as we move away from the origin. To keep things simple, we ignore the ion motion due to the mismatch in inertia I mentioned before, so there is a uniform background of positive ions in which electrons are free to move about. In this case, Poisson’s equation (Gauss’ law with ![]() ) becomes
) becomes
(2) ![]()
We will set the density far away from the origin to ![]() . Assuming that the plasma is in thermal equilibrium, the electron distribution function is of the Boltzmann type;
. Assuming that the plasma is in thermal equilibrium, the electron distribution function is of the Boltzmann type;
![]()
If we integrate this distribution over all speeds and note that as ![]() ,
, ![]() , we can obtain the electron density in terms of the potential
, we can obtain the electron density in terms of the potential ![]() :
:
![]()
We substitute this into Poisson’s equation 2;
![]()
As we discussed earlier, the potential is going to fall quite fast as we move away from the origin, so we can safely assume ![]() and expand the exponential in a Taylor series. We will drop the second order term so that
and expand the exponential in a Taylor series. We will drop the second order term so that
![]()
leading to a solution of exponential decay;
![]()
This defines a length scale which is called the Debye length:
(3) ![]()
This distance can be understood as a characteristic shielding length in a plasma. If the Debye length is sufficiently small (in other words, if the characteristic length of your experimental scenario is ![]() ,
, ![]() ), then any local disruptions of the charge balance in the plasma will be shielded away from the macro perspective; i.e. the plasma can be considered quasi-neutral. Of course, we need to have a plasma density high enough such that the number of charges in the Debye sphere is large,
), then any local disruptions of the charge balance in the plasma will be shielded away from the macro perspective; i.e. the plasma can be considered quasi-neutral. Of course, we need to have a plasma density high enough such that the number of charges in the Debye sphere is large, ![]() .
.
Another important characteristic quantity of a plasma is the plasma frequency ![]() . Suppose you have a slab of plasma for which all the electrons in an interval
. Suppose you have a slab of plasma for which all the electrons in an interval ![]() are shifted one
are shifted one ![]() in the positive direction, resulting in an electric field at
in the positive direction, resulting in an electric field at ![]() . If we use the integral form of Gauss’ law with one boundary of the integration surface at
. If we use the integral form of Gauss’ law with one boundary of the integration surface at ![]() with surface area
with surface area ![]() , and the other boundary far off at large
, and the other boundary far off at large ![]() where the plasma is neutral, then we can come up with an expression for the magnitude of the electric field as a function of the displacement;
where the plasma is neutral, then we can come up with an expression for the magnitude of the electric field as a function of the displacement;

We know that the equation of motion for a single electron is ![]() , so then the equation that describes the dynamics of the displaced electrons in the plasma is
, so then the equation that describes the dynamics of the displaced electrons in the plasma is
![]()
which denotes an oscillation with frequency
(4) 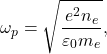
known as the plasma frequency. Another key criterion for plasma is that the frequency of collisions between charged particles and neutral atoms ![]() is small compared to the plasma frequency,
is small compared to the plasma frequency, ![]() . This ensures that the dynamics of the plasma do not end up most closely resembling plain-old hydrodynamics.
. This ensures that the dynamics of the plasma do not end up most closely resembling plain-old hydrodynamics.
This article is an excerpt from Prof. Rodríguez’s PhD thesis. If you would like to cite it for your own work, please cite:
– J. A. Rodríguez, Electromagnetic Wave Manipulation with Plasma Metamaterials, Ph.D. thesis, Stanford University (2023).
[1]: Walter G. Vincenti and Jr. Charles H. Kruger. Introduction to Physical Gas Dynamics. Krieger Publishing, Malabar, FL, 1986.
[2]: Francis F. Chen. Introduction to Plasma Physics and Controlled Fusion, Third Edition. Springer Publishing, Switzerland, 2015.
